- Home
- Medical news & Guidelines
- Anesthesiology
- Cardiology and CTVS
- Critical Care
- Dentistry
- Dermatology
- Diabetes and Endocrinology
- ENT
- Gastroenterology
- Medicine
- Nephrology
- Neurology
- Obstretics-Gynaecology
- Oncology
- Ophthalmology
- Orthopaedics
- Pediatrics-Neonatology
- Psychiatry
- Pulmonology
- Radiology
- Surgery
- Urology
- Laboratory Medicine
- Diet
- Nursing
- Paramedical
- Physiotherapy
- Health news
- Fact Check
- Bone Health Fact Check
- Brain Health Fact Check
- Cancer Related Fact Check
- Child Care Fact Check
- Dental and oral health fact check
- Diabetes and metabolic health fact check
- Diet and Nutrition Fact Check
- Eye and ENT Care Fact Check
- Fitness fact check
- Gut health fact check
- Heart health fact check
- Kidney health fact check
- Medical education fact check
- Men's health fact check
- Respiratory fact check
- Skin and hair care fact check
- Vaccine and Immunization fact check
- Women's health fact check
- AYUSH
- State News
- Andaman and Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Assam
- Bihar
- Chandigarh
- Chattisgarh
- Dadra and Nagar Haveli
- Daman and Diu
- Delhi
- Goa
- Gujarat
- Haryana
- Himachal Pradesh
- Jammu & Kashmir
- Jharkhand
- Karnataka
- Kerala
- Ladakh
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Manipur
- Meghalaya
- Mizoram
- Nagaland
- Odisha
- Puducherry
- Punjab
- Rajasthan
- Sikkim
- Tamil Nadu
- Telangana
- Tripura
- Uttar Pradesh
- Uttrakhand
- West Bengal
- Medical Education
- Industry
Dupilumab Cuts Itch and Hives in Chronic Spontaneous Urticaria: Phase 3 CUPID Trials
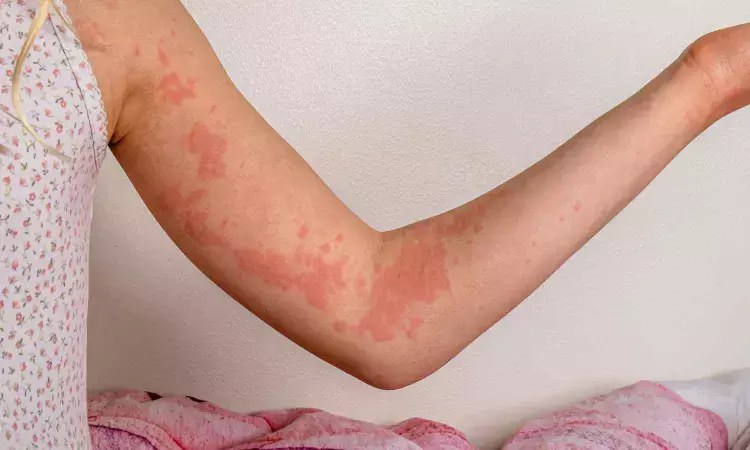
USA: Dupilumab significantly reduced itch and hive severity in patients with chronic spontaneous urticaria (CSU) who remained symptomatic despite antihistamine therapy, findings from the phase 3 LIBERTY-CSU CUPID trials published in JAMA Dermatology have shown. The study was led by Thomas B. Casale from the Division of Allergy and Immunology at the University of South Florida, Tampa, and international collaborators.
- At 24 weeks, dupilumab led to significantly greater reductions in urticaria symptoms compared with placebo.
- The mean reduction in 7-day Itch Severity Score (ISS7) was 8.64 points with dupilumab versus 6.10 points with placebo.
- The 7-day Urticaria Activity Score (UAS7) decreased by 15.86 points in the dupilumab group compared with 11.21 points in the placebo group.
- Pooled analysis of CUPID-A and CUPID-C trials (289 participants) showed consistent improvements in itch severity and overall urticaria activity with dupilumab.
- Symptom improvement with dupilumab was observed as early as week 3 in the combined analysis.
- The safety profile of dupilumab was consistent with previous data, with similar rates of treatment-emergent adverse events in both the dupilumab and placebo groups.
Dr Kamal Kant Kohli-MBBS, DTCD- a chest specialist with more than 30 years of practice and a flair for writing clinical articles, Dr Kamal Kant Kohli joined Medical Dialogues as a Chief Editor of Medical News. Besides writing articles, as an editor, he proofreads and verifies all the medical content published on Medical Dialogues including those coming from journals, studies,medical conferences,guidelines etc. Email: drkohli@medicaldialogues.in. Contact no. 011-43720751


